The journey to ISO 9001 certification is a significant undertaking, and the iso 9001 stage 1 audit often feels like the first major hurdle. The process can seem bureaucratic and overwhelming, leaving many businesses unsure which documents are critical and anxious about potential delays in their certification timeline. This initial feeling of uncertainty is common, but it can be replaced with structured preparation and confidence.
This comprehensive guide is designed to provide just that. We will demystify the Stage 1 audit, providing a clear, methodical roadmap to navigate this crucial documentation review. Inside, you will find an actionable preparation checklist, expert insights into what auditors are looking for, and proven strategies to avoid common pitfalls. Our goal is to ensure you meet all requirements and proceed to your final certification audit with a successful outcome, ready to achieve compliance with confidence.
Key Takeaways
- Understand the Stage 1 audit’s purpose as a readiness review, allowing you to identify and address system gaps before the final certification assessment.
- Discover the key documents and quality management system (QMS) structures an auditor prioritizes to verify your system is designed for compliance.
- Implement a methodical preparation plan to avoid common findings, ensuring your iso 9001 stage 1 audit confirms your readiness for certification.
- Learn how to interpret the audit report and findings to create a clear, actionable path toward a successful Stage 2 certification.
What is an ISO 9001 Stage 1 Audit? (And Why It Matters)
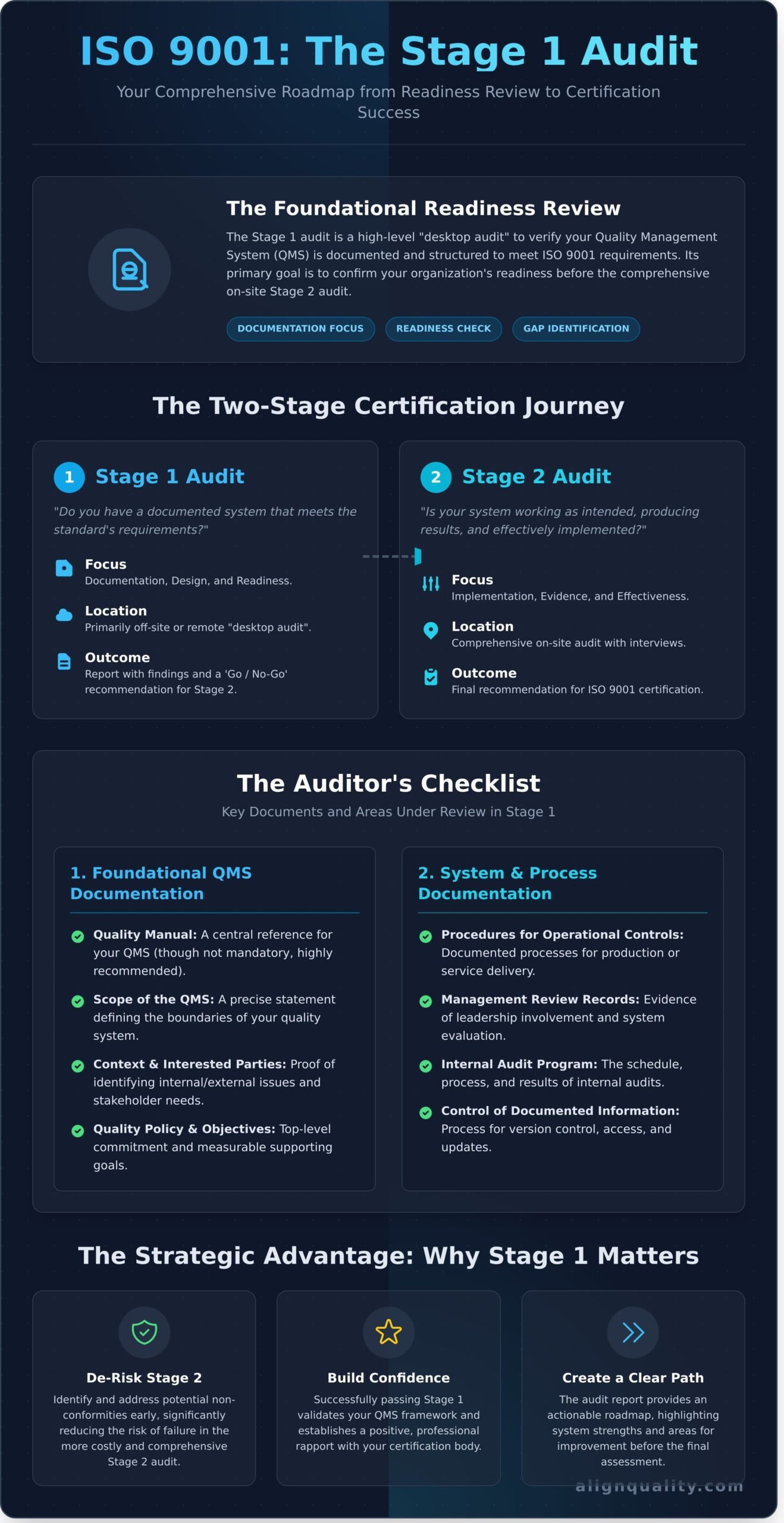
The ISO 9001 certification process is a methodical journey divided into two distinct parts. The ISO 9001 Stage 1 audit serves as the critical first step, acting as a foundational readiness review of your Quality Management System (QMS). It is not a detailed examination of compliance but rather a high-level assessment to determine if your system is structured to meet the standard’s requirements.
Think of it as a “desktop audit.” The auditor primarily reviews your core QMS documentation, such as your quality manual, procedures, and objectives. This evaluation is often conducted partially or entirely off-site. Its purpose is to confirm that you have a documented system in place before proceeding to the more intensive Stage 2 audit, which assesses the actual implementation and effectiveness of that system. This initial step is a vital opportunity to identify potential gaps and non-conformities, allowing you to address them proactively.
The Strategic Goal: From Readiness Check to Risk Reduction
The primary goal of the Stage 1 audit is to verify that the design of your QMS is sound and addresses all applicable clauses of the standard. The auditor confirms that your organization understands its scope, context, and any statutory and regulatory requirements. This review ensures your system is built on the core principles of the ISO 9000 family of standards. By validating your framework upfront, this audit significantly de-risks the more costly and comprehensive Stage 2 audit. Successfully passing Stage 1 builds confidence and establishes a positive rapport with your certification body, setting a professional tone for the remainder of your certification journey.
Stage 1 vs. Stage 2 Audit: The Key Differences
Understanding the distinction between the two audit stages is essential for effective preparation. While they are connected, their objectives, methods, and outcomes are fundamentally different.
- Focus: Stage 1 concentrates on documentation, design, and readiness. The auditor asks, “Do you have a documented system that meets the standard?” In contrast, Stage 2 focuses on implementation, evidence, and effectiveness, asking, “Is your system working as intended and producing results?”
- Location: Stage 1 can often be conducted remotely or with minimal on-site presence. Stage 2 is a comprehensive on-site audit, involving interviews with staff, observation of processes, and a review of records.
- Outcome: The Stage 1 audit results in a report detailing strengths and areas for improvement, culminating in a ‘go’ or ‘no-go’ recommendation for Stage 2. The outcome of the Stage 2 audit determines whether your organization will be recommended for ISO 9001 certification.
The Auditor’s Checklist: Key Documents and Areas Under Review
The primary objective of the iso 9001 stage 1 audit is not to find non-conformities, but to verify that your Quality Management System (QMS) is documented, structured correctly, and ready for a full assessment. The auditor acts as a methodical partner, reviewing your documentation to confirm that you have defined the scope of your certification accurately and established the key processes required by the standard. This comprehensive review forms the strategic plan for the on-site Stage 2 audit.
Foundational QMS Documentation
An auditor begins by examining the high-level framework of your QMS. They need to see a clear and logical structure that defines what your system covers and why. This foundational review confirms your strategic direction and commitment to quality. Key documents include:
- Quality Manual: While no longer mandatory, a well-structured manual is highly recommended as a central reference for your QMS.
- Scope of the QMS: A precise statement defining the boundaries of your QMS, including any justified exclusions.
- Context and Interested Parties: Documentation showing you have identified internal and external issues and the needs of stakeholders.
- Quality Policy and Objectives: Your organization’s top-level commitment to quality and the measurable objectives that support it.
System and Process Documentation
Next, the auditor verifies that your foundational policies are supported by practical, documented processes. This is where the theoretical framework of your QMS connects with day-to-day operations. The auditor looks for evidence that the system is not just planned, but actively functioning through documents such as:
- Procedures for key operational controls (e.g., production, service delivery).
- Records from management reviews, demonstrating leadership involvement.
- Your internal audit program, including the schedule and recent results.
- The process for controlling documented information (e.g., version control, access).
Site and Readiness Evaluation
Finally, the iso 9001 stage 1 audit includes a high-level review of your physical environment and team’s preparedness. The auditor confirms that the documented QMS aligns with the reality of your operations and that your organization is ready for the next step. This evaluation includes a review of site-specific conditions, confirmation of resource allocation, and brief discussions with key personnel to determine overall readiness for the Stage 2 on-site audit.
Your Step-by-Step Plan for Stage 1 Audit Preparation
A methodical approach is the key to navigating your certification journey with confidence. Treating preparation for the iso 9001 stage 1 audit as a formal project eliminates last-minute stress and ensures no detail is overlooked. This structured plan transforms a potentially overwhelming task into a series of manageable milestones. We recommend beginning this process at least 3-6 months after you start implementing your Quality Management System (QMS). Appoint a dedicated internal audit coordinator to manage the timeline, assign responsibilities, and serve as the central point of contact for all audit-related activities.
Phase 1: Internal Verification (3 Months Out)
This initial phase is your opportunity for critical self-assessment and remediation. The objective is to identify and resolve any non-conformities before the external auditor arrives, demonstrating a proactive commitment to quality. A rigorous internal review validates that your QMS documentation and implementation are aligned with the standard’s requirements.
- Conduct a thorough internal audit: Systematically review your processes and records against every applicable ISO 9001 clause.
- Perform a gap analysis: Identify any missing policies, procedures, or records required by the standard. To ensure a comprehensive review, consider using an expert-led gap analysis checklist guide.
- Implement corrective actions: Address all findings with documented corrective actions, proving your system’s capacity for continuous improvement.
Phase 2: Document Collation & Review (1 Month Out)
With your internal findings addressed, the focus shifts to organizing the evidence for the auditor. The Stage 1 audit is primarily a documentation review, so clear, complete, and accessible records are critical to success. This is your chance to present your QMS professionally and make a strong first impression on the auditor.
- Centralize documentation: Create a single, logically organized folder (digital or physical) containing all required QMS documents, from your Quality Manual to process records.
- Review and refine: Scrutinize every document for accuracy, proper version control, and clarity. Ensure documents are approved and formally released.
- Train your team: Confirm that key personnel know exactly where to find relevant procedures and can speak confidently about their roles within the QMS.
Phase 3: Final Checks (1 Week Out)
In the final week, your efforts turn to logistics and team readiness. A smooth, well-coordinated audit day reflects positively on your organization’s management capabilities and overall preparedness. These final preparations ensure there are no surprises, allowing your team to approach the iso 9001 stage 1 audit with composure.
- Confirm logistics: Liaise with the auditor to confirm the schedule, attendees, and any technology needed for remote access.
- Brief your team: Hold a pre-audit meeting to review the audit plan, discuss what to expect, and advise on how to answer questions directly and honestly.
- Prepare the environment: Ensure the meeting space is ready and that all necessary equipment is tested and functional.
Common Stage 1 Pitfalls and How to Avoid Them
Awareness is the most effective form of prevention. Many findings during an ISO 9001 stage 1 audit are not related to operational failures but to incomplete or poorly defined systems. The auditor is primarily assessing if your Quality Management System (QMS) is designed correctly and ready for the main audit. They are looking for disconnects between your stated policies, quality objectives, and the processes you have documented.
Identifying any of these issues is manageable, but receiving a ‘major non-conformance’ at this stage is a serious setback. It indicates a fundamental failure in the system’s design and will halt the certification process until significant corrective action is taken and verified. Avoiding these common pitfalls is critical to ensuring your audit journey proceeds smoothly.
Inadequate Documentation and Scope
A common pitfall is a QMS scope that is poorly defined, inaccurate, or fails to cover all relevant business activities. To avoid this, clearly document the physical locations, processes, and departments your QMS covers and provide a clear justification for any clauses of the standard you have excluded. Another frequent issue is missing key documents required by the standard. The solution is to use a checklist to systematically verify that all mandatory documented information is in place and approved.
Weak Internal Audit & Management Review
Auditors will immediately look for evidence of a robust internal quality cycle. A superficial internal audit that only scratches the surface is a significant red flag. Ensure your internal audit is comprehensive, covers all relevant clauses of ISO 9001, and is conducted by a competent and impartial auditor. Likewise, a lack of formal evidence of a management review is a critical error. You must conduct and thoroughly minute at least one management review before the Stage 1 audit, documenting all inputs, discussions, and resulting action items.
Failure to Address System Gaps
Ignoring known weaknesses in your QMS is a recipe for audit findings. An auditor will appreciate transparency and proactive management far more than the discovery of a hidden issue. Proactively identify gaps through your internal audits and risk assessments, and demonstrate that a plan is in place to correct them. It is also vital to have a clear understanding of how to manage findings. Learning to effectively document and resolve potential ISO 9001 nonconformances before the audit demonstrates system maturity and control.
After the Audit: Understanding Outcomes and Next Steps
The conclusion of your ISO 9001 Stage 1 audit is marked by a formal closing meeting. Here, the auditor will present a summary of their findings, provide a recommendation for the next phase, and issue a written report. This meeting is your final opportunity to ask for clarification on any points raised, ensuring you have a clear understanding of your Quality Management System’s standing.
The auditor’s recommendation will fall into one of three categories. This outcome is critical, as it directly determines your timeline and the necessary actions required to proceed to the Stage 2 certification audit.
Outcome 1: Recommended for Stage 2
This is the ideal result, indicating your QMS documentation and design are fundamentally sound. The auditor has found no major roadblocks and will recommend proceeding. The Stage 2 audit is typically scheduled 4 to 8 weeks later, giving you time to address any minor ‘Areas for Improvement’ noted in the report and consolidate your system for the on-site evaluation.
Outcome 2: Minor Non-conformances Found
It is common for an auditor to identify minor non-conformances. These are small gaps in your system that do not represent a systemic failure. To move forward, you must submit a formal corrective action plan detailing how you will resolve each finding. Once your certification body accepts the plan, the Stage 2 audit can be scheduled. View this outcome as constructive feedback to strengthen your QMS.
Outcome 3: Major Non-conformance Found
A major non-conformance indicates a significant failure to meet a requirement of the ISO 9001 standard. This finding will pause your certification journey until the issue is fully resolved and verified. A follow-up audit may be required to confirm the effectiveness of your corrective actions. This outcome highlights why thorough preparation is vital to the overall ISO certification process.
Understanding the results of your iso 9001 stage 1 audit is the first step toward successful certification. Each outcome provides a clear path forward. By methodically addressing the auditor’s findings, you can approach your Stage 2 audit with confidence and ensure your quality management system is robust and effective.
Navigate Your Stage 1 Audit with Confidence
The iso 9001 stage 1 audit is a critical milestone on your certification journey, serving not as a final exam, but as a collaborative review of your QMS documentation and overall readiness. As this guide has demonstrated, success is the direct result of meticulous preparation, a clear understanding of the auditor’s requirements, and a proactive approach to addressing potential nonconformities. Mastering these elements sets a powerful foundation for a smooth and successful Stage 2 audit.
However, you do not have to navigate this complex process alone. Align Quality provides the expert partnership necessary to streamline your preparation and build unshakeable confidence. Guided by our team of Certified ISO 9001 Lead Auditors, who bring extensive multi-industry experience, our proven 5-Stage Certification Process removes uncertainty and ensures you meet every requirement with precision and clarity.
Ensure Stage 1 success. Book a free consultation with our experts.
Take the next step with a partner dedicated to your success. Your successful certification is well within reach.
Frequently Asked Questions About the ISO 9001 Stage 1 Audit
How long does an ISO 9001 Stage 1 audit typically take?
The duration of an ISO 9001 Stage 1 audit is typically one to two days. However, this timeframe is dependent on several key factors, including the size of your organization, the complexity of your operations, and the number of physical locations included in your Quality Management System (QMS) scope. Your certification body will provide a precise audit plan with a confirmed duration based on their initial assessment of your organization’s structure and requirements.
Can the Stage 1 audit be conducted entirely remotely?
Yes, it is increasingly common for the Stage 1 audit to be conducted remotely. This stage primarily involves a review of your QMS documentation, which can be shared and discussed effectively through virtual meetings and secure platforms. The final decision rests with your certification body, which will assess feasibility based on your organization’s complexity. A remote audit offers significant flexibility and efficiency, streamlining the initial phase of your certification journey with confidence.
What happens if we fail the Stage 1 audit?
A Stage 1 audit is a readiness review, so it is not a pass/fail event in the traditional sense. If the auditor identifies significant issues or non-conformances, they will be detailed in an audit report. Your organization will then have a defined period to implement corrective actions to address these findings. The Stage 2 audit will not be scheduled until these critical issues are resolved, ensuring your QMS is robust enough for the full certification assessment.
Do we need to have our entire QMS fully implemented for several months before Stage 1?
No, full implementation for an extended period is not a prerequisite for Stage 1. This initial audit focuses on verifying that your QMS is well-documented and designed to meet the ISO 9001 standard. While key components like your quality policy and objectives should be established, the primary goal is to confirm readiness. Evidence of full, long-term implementation and the generation of records become critical for the subsequent Stage 2 certification audit, not for Stage 1.
What is the difference between a ‘minor’ and ‘major’ non-conformance in a Stage 1 audit?
In an iso 9001 stage 1 audit, a major non-conformance signifies a systemic failure, such as the complete absence of a required procedure or a breakdown that could prevent meeting customer requirements. A minor non-conformance is an isolated lapse or a single deviation from a defined process. Major findings must be fully resolved before you can proceed to the Stage 2 audit, ensuring foundational issues are corrected first, while a plan for minors is often sufficient.
Is it possible to combine the Stage 1 and Stage 2 audits?
Combining the Stage 1 and Stage 2 audits is highly uncommon and generally not recommended. The two stages serve distinct purposes: Stage 1 is a readiness review, while Stage 2 is a detailed assessment of implementation. The gap between them provides a crucial opportunity to address any findings from Stage 1. While a certification body might consider a combined audit for a very small, simple organization, the standard, two-stage approach is the proven methodology for success.
Who from our company should attend the Stage 1 audit meetings?
Key personnel should be present to demonstrate management commitment and provide comprehensive information. This typically includes top management (e.g., CEO, General Manager), the appointed Quality Manager or Management Representative, and the heads of key departments whose processes are central to the QMS. Their participation ensures the auditor can gain a clear, high-level understanding of your system’s design, objectives, and the resources allocated to maintain quality and compliance throughout the organization.